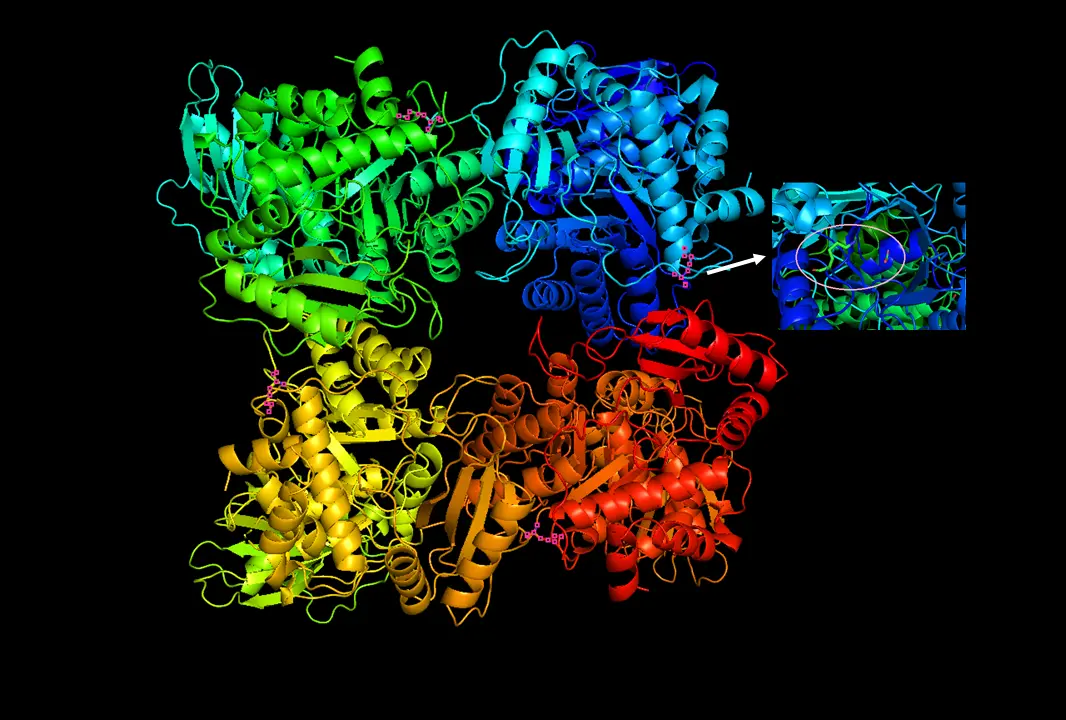
RuBisCO
4 billion years of fixing inorganic carbon in the biosphere. Sometimes mistakes O2 for CO2. Not as fast as some enzymes, but very abundant. Here, have some phosphoglycerates about it.
- 0 Posts
- 21 Comments
Love it. One example that springs to mind is calling out the NCAA.
“‘Student Ath-uh-letes’? Haha, that is brilliant, sir.”
Holy shit, the unburned powder on the ballistics gel block!
This one should bookend that rifle with the crazy long barrel they keep sawing pieces off of while monitoring muzzle velocity.
Oh we’re all going nocturnal with this climate. It will be necessary. Maybe subterranean, too.
 74·2 months ago
74·2 months agoYour jncos may be frayed and soaked, but the Slim Shady LP just came out and you’re off to the theater with friends to see The Matrix.

 3·2 months ago
3·2 months agoIt’s a good podcast, I recommend it to anyone who likes plants. The host is a non-traditional student who swears a lot, very
New Jersey/New YorkChicago.https://www.outsideonline.com/culture/books-media/botany-joey-santore-youtube-crime-pays/
Totally understandable, so do I.

 2·3 months ago
2·3 months agoiNaturalist?! I wanna see these pics! Do you like Crime Pays But Botany Doesn’t?

 6·3 months ago
6·3 months ago- Easy access to dark skies for stargazing.

 8·3 months ago
8·3 months agoPesticides
It’s shooting like this that’ll cost you a pinky finger in the long run.

Ohh yeahh! Now I got you.
Those were wild times. You had to be there.

 2·4 months ago
2·4 months agoI could also be wrong, but I believe SDS has less ‘affinity’ for protons than acetic acid (which is part of the reason why detergents work so well). You’d need sulfuric acid, or something stronger, and removal from solution of its buddy ion sodium. Then I think you could protonate dodecyl sulfate.
Now acetic acid and soaps…yeah, far more likely to generate scum. The polar head is a weaker acid.
The importance of soap to human civilization is documented by history, but some problems associated with its use have been recognized. One of these is caused by the weak acidity (pKa ca. 4.9) of the fatty acids. Solutions of alkali metal soaps are slightly alkaline (pH 8 to 9) due to hydrolysis. If the pH of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum. A second problem is caused by the presence of calcium and magnesium salts in the water supply (hard water). These divalent cations cause aggregation of the micelles, which then deposit as a dirty scum.
These problems have been alleviated by the development of synthetic amphiphiles called detergents (or syndets). By using a much stronger acid for the polar head group, water solutions of the amphiphile are less sensitive to pH changes. Also the sulfonate functions used for virtually all anionic detergents confer greater solubility on micelles incorporating the alkaline earth cations found in hard water.
Oh wow, the book was based on the movie? That explains some things.

 30·4 months ago
30·4 months ago“Howdy, XO,” he drawled. The old west affectation common to everyone from the Mariner Valley annoyed Holden. There hadn’t been a cowboy on Earth in a hundred years, and Mars didn’t have a blade of grass that wasn’t under a dome, or a horse that wasn’t in a zoo. Mariner Valley had been settled by East Indians, Chinese, and a small contingent of Texans. Apparently, the drawl was viral. They all had it now. “How’s the old warhorse today?”
James S.A. Corey, Leviathan Wakes

 1·5 months ago
1·5 months agoLooking very Shane today.
Alabama Shakes - Sound and Color

 15·6 months ago
15·6 months agoWhen things get really tough, two of you will double up on the same keyboard.
1 in 6 have multiple personalities and substance abuse daemons.
Your bosses ride little skateboards everywhere, when they’re not busy programming animated singing viruses.
The FBI watches you code, but has no idea what they’re looking at.
A significant fraction of you can type with your feet, proficiently.




https://en.wikipedia.org/wiki/Jim_Henson
https://en.wikipedia.org/wiki/Streptococcus_pneumoniae
https://en.wikipedia.org/wiki/Streptococcus_pyogenes
https://en.wikipedia.org/wiki/The_Walt_Disney_Company